Fluorescence excitation and emission are like the dazzling stars of the molecular light show. In this friendly exploration, we’ll uncover the magic of how molecules absorb energy, get excited, and then radiate light. These processes play a pivotal role in various scientific fields, from biology to materials science.
Think of it as a dance of molecules in the spotlight, where they absorb specific colors of light and then return the favor by emitting their vibrant hues. It’s a fascinating world where we learn how to choose the right colors, actors (fluorophores), and spotlights (light sources) for this captivating molecular performance. So, let’s embark on this illuminating journey together!
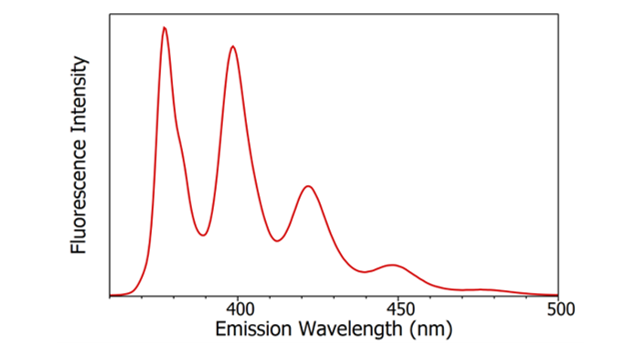
Fluorescence Excitation
Fluorescence is a fascinating world of light and molecules that scientists explore every day. But how does it all begin? Let’s start into the basics of fluorescence excitation in a friendly and easy-to-understand way.
Absorption of Light: Think of molecules as tiny sponges that soak up light. They do this through a process called absorption. Each molecule has its favorite colors, and when they absorb those colors, they get excited!
Absorption Spectra: Imagine an absorption spectrum as a molecule’s unique fingerprint. It shows us the colors it likes to absorb. Scientists use this fingerprint to recognize and study molecules, just like we recognize people by their clothing.
Beer-Lambert Law: This law is like a rulebook for understanding how much light molecules can absorb. It tells us about concentration, path length, and absorbance. It’s simple, like knowing the capacity of a sponge.
Role of Absorption in Excitation: So, what happens when molecules absorb light? They get excited, like going from a quiet day to a party night. This excitement is the key to fluorescence – making molecules emit light.
Selection of Excitation Wavelength: Choosing the right color of light is crucial. Scientists pick an excitation wavelength that matches the absorption peak, like using the right key for a door. This ensures maximum excitement!
Fluorophores and Excitation Sources: Now, let’s meet our stars – fluorophores! These are special molecules that can fluoresce, or glow. Picking the right fluorophore is like casting the perfect actor for a role.
Fluorophore Selection: Think of fluorophores as actors in a movie. Scientists pick them based on properties like brightness, stability, and color, just like choosing actors for a blockbuster film.
Light Sources (Lasers, Lamps, LEDs): Fluorophores need a spotlight to shine. Light sources, such as lasers, lamps, and LEDs, provide this spotlight. It’s like selecting the perfect stage lighting for our actors.
Fluorescence excitation is like preparing for an amazing performance. We choose the right colors, actors (fluorophores), and spotlights (light sources) to make molecules dazzle. By understanding absorption, absorption spectra, and the Beer-Lambert Law, we ensure our molecules are in party mode, ready to emit light.
Fluorescence Emission
Emission spectra are like a dazzling light show that molecules put on after absorbing energy. In this friendly journey through the world of fluorescence, we’ll explore the significance of the Stokes Shift, the concept of Fluorescence Lifetime, the Fluorescence Quantum Yield, and the various factors that affect emission.
Stokes Shift and Its Significance: Imagine a molecule as a spring. When it absorbs energy and gets excited, it stretches like a spring wound up. But when it relaxes and emits light, it doesn’t go back to its original position; it stops short. This energy difference is the Stokes Shift, and it’s crucial because it tells us something exciting happened!
Fluorescence Lifetime: Now, let’s delve into Fluorescence Lifetime. Think of it as a molecule’s dance move. It measures how long a molecule can stay in its excited state before returning to its normal state. Scientists measure this dance move to understand what’s happening at the molecular party.
Definition and Measurement: Fluorescence Lifetime is like counting the beats of music. Instruments can precisely measure how long a molecule stays excited. This helps scientists figure out what the molecule is up to, whether it’s mingling with others or doing its dance.
Applications: Fluorescence Lifetime isn’t just for fun; it has important applications. For example, in biology, it helps researchers study how molecules interact in living cells. It’s like peeking into a busy dance floor to see who’s dancing with whom.
Fluorescence Quantum Yield: This term measures how efficient a molecule is at emitting light after getting excited. Think of it as the ratio of dancers who show off their moves at a party. A high quantum yield means lots of bright dancers, while a low one means the party is dim.
Factors Affecting Emission: Emission can be affected by various factors, just like a party can change depending on the atmosphere, drinks, and music. Let’s look at these factors:
Environmental Factors: The surroundings play a big role. Temperature, pH, and the presence of other molecules can affect how brightly a molecule emits light. It’s like how the mood at a party can change with the room temperature and crowd.
Chemical Factors: The molecule itself can influence its emission. Chemical changes like oxidation or interactions with other molecules can alter its dance moves. It’s like a dancer changing their routine based on their partner.
Instrumentation Factors: The tools we use to measure fluorescence matter too. The sensitivity and accuracy of instruments can impact the quality of data we get. It’s like having the right dance floor and music for a perfect performance.
Emission spectra are like the grand finale of the fluorescence show. Understanding the Stokes Shift, Fluorescence Lifetime, and Fluorescence Quantum Yield helps scientists decode the molecular dance happening at the atomic level. It’s a world of colorful afterglows and scientific discovery, where molecules reveal their secrets with every dance move.
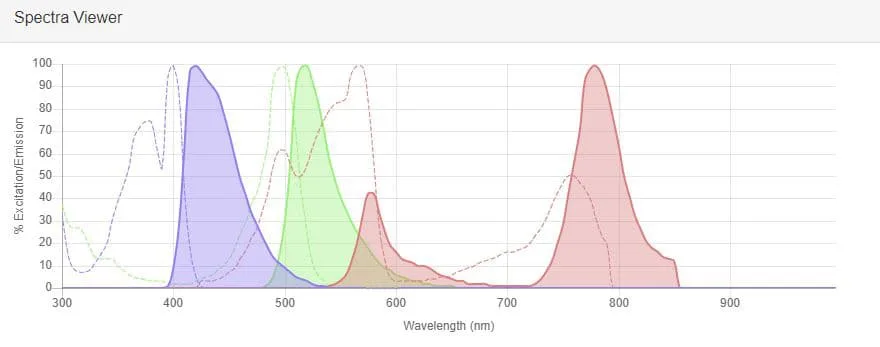
What is the difference between fluorescence and emission?
Fluorescence and emission are like two sides of the same vibrant coin in the world of light and molecules. Let’s uncover the friendly differences between them.
Fluorescence is a type of emission, but it has some unique characteristics. When we talk about fluorescence, we usually mean the emission of light that happens almost immediately after a molecule absorbs energy. It’s like a quick and dazzling response to a stimulus.
Emission, on the other hand, is a broader term. It encompasses all instances where a molecule releases energy in the form of light. This includes fluorescence but also other processes like phosphorescence, chemiluminescence, and bioluminescence. It’s like a big family of light emission events.
The key distinction is in the timing. Fluorescence is like a rapid “flash” of light, while emission covers a wider range of light-release scenarios. So, when you think of fluorescence, picture a swift and eager reaction, whereas emission is the larger, inclusive concept that embraces all the ways molecules can shine their radiant light upon the world.
Final Words
Fluorescence excitation and emission are like the dynamic duo of molecular storytelling. They unveil the hidden tales of molecules, allowing us to see their vibrant colors and behaviors.
